The Compliance Guardrail: Maintaining Pharma Brand Consistency in Global Markets
According to a study by Marq, brand consistency delivers real financial impact in a company, lifting revenue from 5 percent up to beyond 20 percent. In today’s competitive landscape, this data shows that design teams should be viewed through a more strategic lens, as their work can directly influence your business performance.
In pharma, creating compliant and effective pharma decks is more complex than in any other industry. With rigorous MLR requirements, both branding consistency and data integrity are non-negotiable.
After +10 years in the market, we realized that when pharma slides are edited locally and fragmented across teams, compliance risk rises, scientific messaging weakens, and Healthcare Professionals (HCPs) trust erodes.
Want to learn more about this topic? Keep reading!
The High Cost of Visual Inconsistency in Pharma Launches
Visual inconsistency is one of the highest invisible costs in the medical industry. During a global launch, the brand can happen fast.
Let’s check some scenarios:
- The design time is high. According to our study, pharma professionals spend an average of 7 hours per presentation, and each slide requires roughly 65 minutes of design time due to the scientific complexity. Not taking that into account, as they aren’t design experts, they are likely to invest more time, which ultimately equates to money.
- HCP engagement may decline. With industry reports showing declining engagement rates, unclear or inconsistent visuals reduce trust and accelerate drop-off among Healthcare Professionals (HCPs).
- A regulatory risk is also possible. Inconsistent pharma presentations can unintentionally alter the display of technical details, increasing the risk of error. Now, amid intensified FDA enforcement, even minor visual imbalances can pose direct regulatory risk.
- Manual changes can lead to regional inconsistencies. It’s common for medical staff and field teams to adjust slides to meet regional needs, and each manual change can introduce new formatting and stylistic inconsistencies.
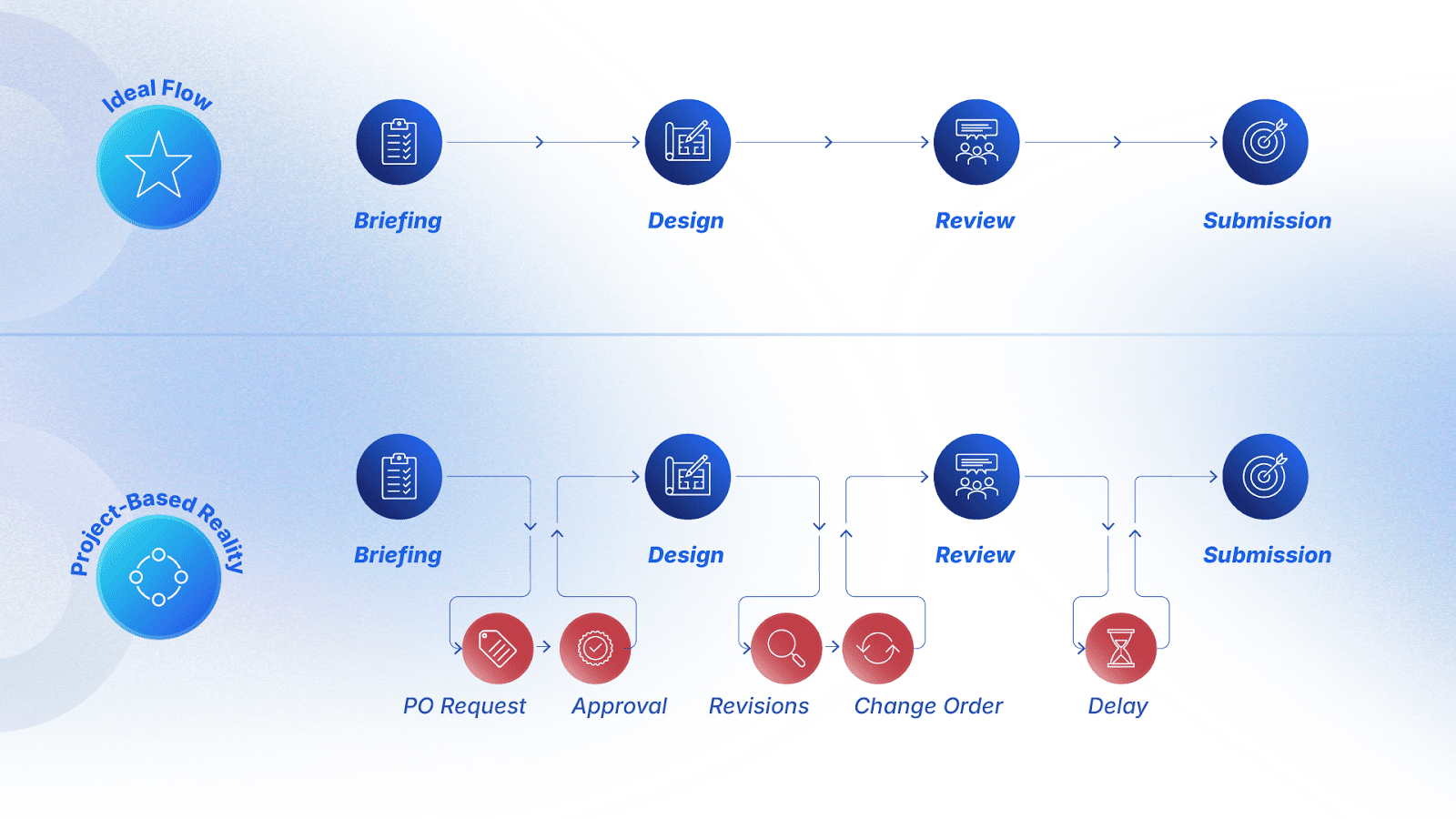
How Manual Slide Design Increases MLR Risk?
At 24Slides, we believe that manual slide design increases MLR review challenges and compliance delays. Let’s see with more details:
- Current visual standards focus on visual clarity. Regulators evaluate font size, contrast, risk proximity, and layout neutrality. It’s natural that annual or local edits often break these requirements.
- MLR auditability suffers when multiple versions circulate. When each affiliate modifies its own pharma visual aid PPT, it becomes difficult to verify if every deck meets legibility and disclosure standards.
- Generative tools cannot interpret regulatory norms. The research shows that AI tools often break brand rules, distort hierarchy, or omit mandatory visual elements. The responsibility for fixing these issues lies with internal teams.
Manual formatting in pharma slides is not only inefficient. It also creates more compliance touchpoints, more review loops, and more risk.
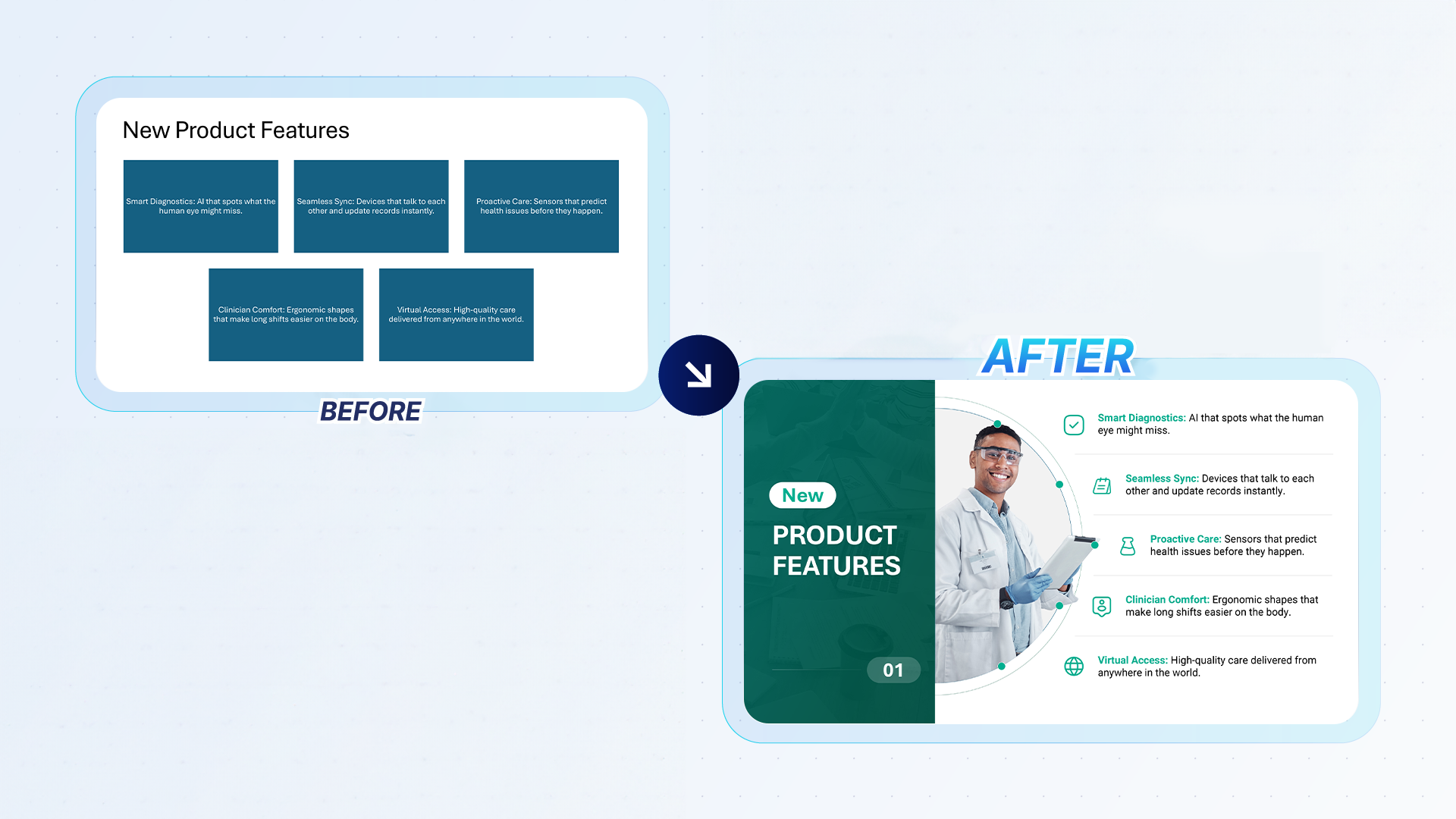
How to Standardize the Regulatory Design Workflow and Your Brand Consistency?
Up to this point, you will have noticed that maintaining brand consistency in the medical field goes beyond a marketing task: it is a strategic task that will determine your business success.
With that in mind, here are some recommendations:
- Embed fair balance in the visual structure. Ensure that risks and benefits are presented with comparable prominence in every deck and slide. This includes using a similar type size, layout, and visual weight.
- Adopt clear, conspicuous, and neutral presentation standards. You must avoid distractions or formatting that could downplay safety content, as required by FDA communication rules.
- Support quantitative accuracy in charts and tables. Adopt internal standards for data visualization that mirror regulatory expectations (e.g., correct axes, full risk-benefit display), reducing the chance that visuals could be interpreted as misleading.
- Integrate submission requirements early. Create design workflows that capture FDA advertising requirements. The recommendation is to prepare and upload the guidelines from the beginning to avoid changes at the end of the design process.
- Train dedicated designers on your corporate visual identity, not generic pharma guidelines. This ensures every asset stays true to your brand universe and regulatory rules, regardless of any change of management.
These standardization practices help ensure regulatory compliance, minimize review cycles, and protect launch timelines, while eliminating guesswork and manual rework.
The Final ROI of MLR-Ready Slides in Pharma
Future-proofing your brand means building design systems that scale across launches, markets, and any change management.
Presentation design in Pharma is not optional; it is inevitable.
- Consistency strengthens HCP trust. When clinicians encounter clear, consistent pharma visual aid materials across touchpoints, credibility increases.
- Time saved is money saved. Leaders spend fewer hours editing slides and more hours on strategic work. The research shows significant salary leakage up to $692K tied to manual formatting and design tasks.
- Regulatory safety is reinforced. Strict adherence to readability and fair balance guidelines lowers the chance of corrective action in an era of heightened FDA scrutiny.
At 24Slides, we bring over 10 years of experience in presentation design, and we believe that a consistent brand system is not a creative luxury, it is an operational and regulatory asset.

The Final Pivot: Ready to Audit Your Compliance Velocity?
See how much time your team is losing to manual formatting and how much capacity you can redirect into strategic, compliant global launches.
For us, it’s important to help you optimize your design operations and elevate your performance like never before.
If you want to learn more about our research and the time you're losing on design tasks, consult the Pharma White Paper!





