Beyond the Pixel: How 24Slides Empowers Pharma Brand Managers to Reclaim Strategic Velocity
In the high-stakes pharmaceutical industry, every hour spent on manual slide production is an hour lost to high-level strategy. Our research into over 1,000 life sciences professionals reveals that medical presentation design is the missing link in pharmaceutical marketing budget optimization.
In this article, we’ll cover:
- Why Presentation Strategy is the New Frontier for Pharma Efficiency: Understanding how specialized design transforms high-stakes slides from a resource drain into a strategic asset.
- Reclaiming the "Salary Leakage" through Medical Presentation Design: Addressing the opportunity cost of the 4.3 hours per week lost by senior directors to manual formatting.
- Beating the 65-Minute-Per-Slide Bottleneck: How professional visualization drives faster speed-to-market by removing internal production hurdles.
- How Visual Clarity Shortens the Path to MLR Approval: Utilizing rapid design support to handle complex pharma charts that otherwise stall regulatory timelines.
- Consistency and Compliance—The Life Sciences Product Slide Standard: Mitigating risk through locked templates and precision in complex MOA (Mechanism of Action) diagrams.
- The Final ROI—Redirecting Hours into High-Level Strategy: Quantifying the impact of redirecting saved management hours back into high-level brand strategy.
Why Presentation Strategy is the New Frontier for Pharma Efficiency
The modern Brand Manager is caught in a "technical squeeze." While expected to execute complex omnichannel launches with leaner teams, they often find themselves acting as manual "data translators."
Every hour spent on a pharmaceutical presentation is high-cost labor lost to high-level strategy. To reach Operational Surplus, leaders must treat their pharmaceutical company presentation pipeline as a technical workflow rather than a creative afterthought. While medical slides require specific compliance, the core principles of impact remain the same; see our guide on How to Design Corporate Presentations for a breakdown of high-performance layout structures.
Closing the "Integration Gap" in Clinical Communications
The efficiency frontier in 2026 isn't just about "better slides"; it’s about ecosystem readiness. A pharmaceutical industry PowerPoint presentation must be built to survive the digital life cycle:
- Platform-Ready Assets: Professional design ensures that reference anchoring and fair-balance placement align with FDA promotional guidance while meeting the technical and structural requirements of Veeva Vault’s validated content platform and IQVIA-aligned data standards used across Veeva systems. In practice, this reduces compliance-related revision cycles and minimizes content “bounce-back” during MLR review.
- Data Visualization Translation: Strategic design transforms raw market intelligence into intuitive, at-a-glance visuals that reduce cognitive load and allow HCPs to recognize meaningful patterns in longitudinal patient data almost instantly. By making complex information easier to interpret, these visuals improve comprehension and decision efficiency.
- Medical-Legal-Regulatory (MLR) Speed: By medical presentation design outsourcing to shorten regulatory review cycles.
- Standardized Submission Themes: Using presentation workflow automation, pharma ensures that every asset, from global congress decks to local sales aids, is technically identical and compliant with global brand standards.
By unbundling technical production from high-overhead strategy agencies, Pharma leaders ensure their assets are "platform-native" for the digital ecosystems governing the modern life sciences industry.
Reclaiming the "Salary Leakage" through Medical Presentation Design
Senior leadership time is a strategic asset, not a clerical resource. Diverting this capital toward administrative formatting creates 'Salary Leakage,' hollowing out the productivity required for true pharmaceutical marketing budget optimization.
The Quantified Cost of "Executive Drag"
When senior leaders spend 4.3 hours per week on pharmaceutical presentation PPT tasks, they lose 12% of their work year to manual production.
- The Industry Benchmark: A scientific presentation requires an average of 65 minutes per slide, nearly double the corporate standard, due to data complexity and regulatory nuance.
- The Financial Impact: Reclaiming these hours enables directors to achieve Operational Surplus and oversee additional Phase III rollouts without adding headcount.
By recognizing that a medical presentation is a strategic asset rather than a clerical burden, firms can move toward a Strategic Bandwidth.
The 65-minute "Scientific Tax" exists because medical data requires a level of nuance that generic software lacks. Understanding the technical divide is critical; see why AI vs. Human Precision: Why Generic Tools Fail the Pharma Test is the deciding factor for regulatory-heavy assets.
Stopping the Intellectual Capital Drain
When a clinical lead is forced to act as a part-time designer, the organization loses the very expertise it hired them to provide. To achieve true pharma brand management efficiency, you must pivot from manual labor to a high-value strategy.
- The Problem: Salary Leakage & Lost Authority High-value clinical leads often squander their "Clinical Authority" on administrative pixel-pushing. This creates a productivity gap where the total cost of employment is wasted on tasks below a leader’s core competency. To measure this loss, organizations must look beyond hourly wages and perform internal audits to calculate the fully burdened labor rate, the true cost of every hour lost to formatting.
- The Solution: Presentation Workflow Automation. By shifting production to an outsourced PowerPoint presentation partner, you implement a workflow automation system for pharma presentations. This removes the technical burden of formatting from your internal team.
- The Result: Strategic Reinvestment. This recovery of human capital allows your most expensive talent to exit the "Executive Drag" of formatting text boxes and refocus on high-impact Phase III rollouts and omnichannel launches.
In the rush to automate, many firms weigh the pros and cons of AI Presentation Makers vs. Professional Designers. For Pharma, the choice often comes down to the necessity of human precision in regulatory environments.
How to beat the 65-Minute-Per-Slide Bottleneck
If your team is trapped formatting shapes instead of analyzing data, you are suffering from "Executive Drag." Transition from manual labor to Strategic Bandwidth with this three-step framework:
- Step 1: Automate Technical Workflows. Stop treating the pharmaceutical presentation PPT as a clerical task. By treating slide production as a technical workflow, you eliminate manual formatting bottlenecks, reduce friction for reviewers, and accelerate your path to MLR-Ready status.
- Step 2: Unbundle Production for 60% ROI Achieve pharmaceutical marketing budget optimization by unbundling design from high-overhead strategy agencies. Redesigning these outsourcing relationships drives a 60% reduction in operational costs, redirecting capital from "pixel-pushing" to patient outcomes.
- Step 3: Scale Output via Professional Outsourcing. Pharma launch cycles are volatile. Use professional presentation outsourcing to scale production instantly during "congress season" peaks. This allows your team to handle massive slide volumes without the long-term liability of increasing internal headcount.
Pro Tip: To see how 1,000+ life sciences professionals are reclaiming their time, explore our full report: The Benefits of External Designers for Pharmaceutical Professionals. The Benefits of External Designers for Pharmaceutical Professionals.
How Visual Clarity Shortens the Path to MLR Approval
In the pharmaceutical sector, the speed of a product launch is often dictated by the efficiency of the Medical, Legal, and Regulatory (MLR) review cycle. A major cause of launch delays is "cognitive friction," which is the mental strain reviewers experience when navigating cluttered tables or inconsistent data.
Professional data visualization transforms raw data into intuitive, scanable assets that allow reviewers to verify endpoints and footnotes immediately.
- Accelerating Speed to Market: When a pharmaceutical company presentation uses clear, professional visuals, compliance becomes a streamlined process rather than a bottleneck. This ensures your Clinical Authority is evident from the first slide.
- Leverage a "Follow-the-Sun" Workflow: By using 24-hour slide deck design support, global teams can submit complex chart redraws for a pharmaceutical poster presentation at 5:00 PM and have MLR-Ready assets ready for submission by 9:00 AM the following day.
- Absorbing Volume Spikes: This high-velocity production model handles the massive presentation for product launch, such as adapting a "Master Compendium" into over 100 localized variants, without burning out your internal staff.
Explore more: From Complex Data to Clear Insights: Visualizing Clinical Trial Success —Learn how to transform raw clinical findings into compliant, executive-ready visuals."
Consistency and Compliance: The Life Sciences Product Slide Standard
Maintaining brand integrity across global markets requires a governed infrastructure for every pharma product slide. In the life sciences sector, a single unapproved edit to a font size or disclaimer can lead to significant regulatory friction. Specialized medical presentation design acts as a primary line of defense.
24Slides serves as a strategic catalyst for your workflow, delivering medical presentation design that converts complex clinical data into submission-ready visuals, effectively shortening the gap between the initial draft and final regulatory approval.
Technical Governance & Regulatory Alignment
- "Locked" for Governance: Professional scientific PowerPoint presentation templates utilize restricted access permissions to prevent unauthorized changes to fonts, legal disclaimers, and footers. As Microsoft Support highlights, using restricted access permissions within PowerPoint is a primary line of defense against unauthorized changes to core brand assets.
- Enforcing FDA Standards: A specialized "fair balance" approach; templates ensure this neutrality isn't lost during the design process.
- Precision in MOA Diagrams: Small visual inaccuracies in Mechanism of Action (MOA) diagrams can distort scientific meaning and create compliance hurdles. Research in PMC emphasizes that discrepancies in medical illustrations can mislead viewers, underscoring the clinical necessity of visual accuracy rather than an aesthetic choice.
- Veeva Vault and IQVIA Readiness: Our workflow is optimized for presentation automation in pharma by aligning with enterprise platforms used across life sciences organizations (IQVIA–Veeva interoperability and partnership). This ensures all files are technically compatible with Veeva Vault’s validated content and data environment, reducing upload friction and supporting faster progression toward MLR-ready review.
By implementing these "Submission-Ready" standards, organizations achieve an Operational Surplus. Teams no longer waste hours in revision loops correcting formatting errors, allowing them to focus on the science that drives market share.
Professional clinical trial data visualization ensures that assets are technically accurate for immediate upload into Veeva Vault. To maintain this level of precision across global teams, see how The Compliance Guardrail: Maintaining Global Brand Consistency in Pharmaceutical Design prevents costly regulatory 'bounce-backs'.
Standardizing with Submission-Ready Themes
By utilizing standardized pharmaceutical presentation templates, teams eliminate the revision loops that delay regulatory transitions. This standardization acts as a critical safeguard, ensuring that every pharma product slide adheres to a unified visual language before it even reaches the desk of a reviewer.
- Eliminating Technical Friction: These themes are built specifically for presentation workflow automation in pharma, removing the manual formatting errors that often trigger multiple rounds of internal feedback.
- Killing Revision Loops: High-performance scientific PowerPoint presentation templates include "protected zones" for claims and safety language, ensuring slides are submission-ready by default. As industry research from Pharmaphorum notes, traditional MLR review is incredibly resource-intensive; standardizing the visual input is one of the fastest ways to reduce the number of review cycles required.
- Protecting Clinical Authority: A pre-approved theme ensures that your medical presentation maintains its Clinical Authority by keeping complex layouts consistent and compliant with Veeva Vault or IQVIA requirements.
- Audit-Ready Peace of Mind: Automated version and date stamps within the template ensure every scientific presentation is easily trackable and historically accurate.
What should be included in a scientific presentation? Beyond the core data, a compliant scientific presentation must include a clear "Fair Balance" slide, consistently formatted references, high-resolution MOA diagrams with standardized labels, and a clear version control system to ensure field teams are always using the latest approved data.
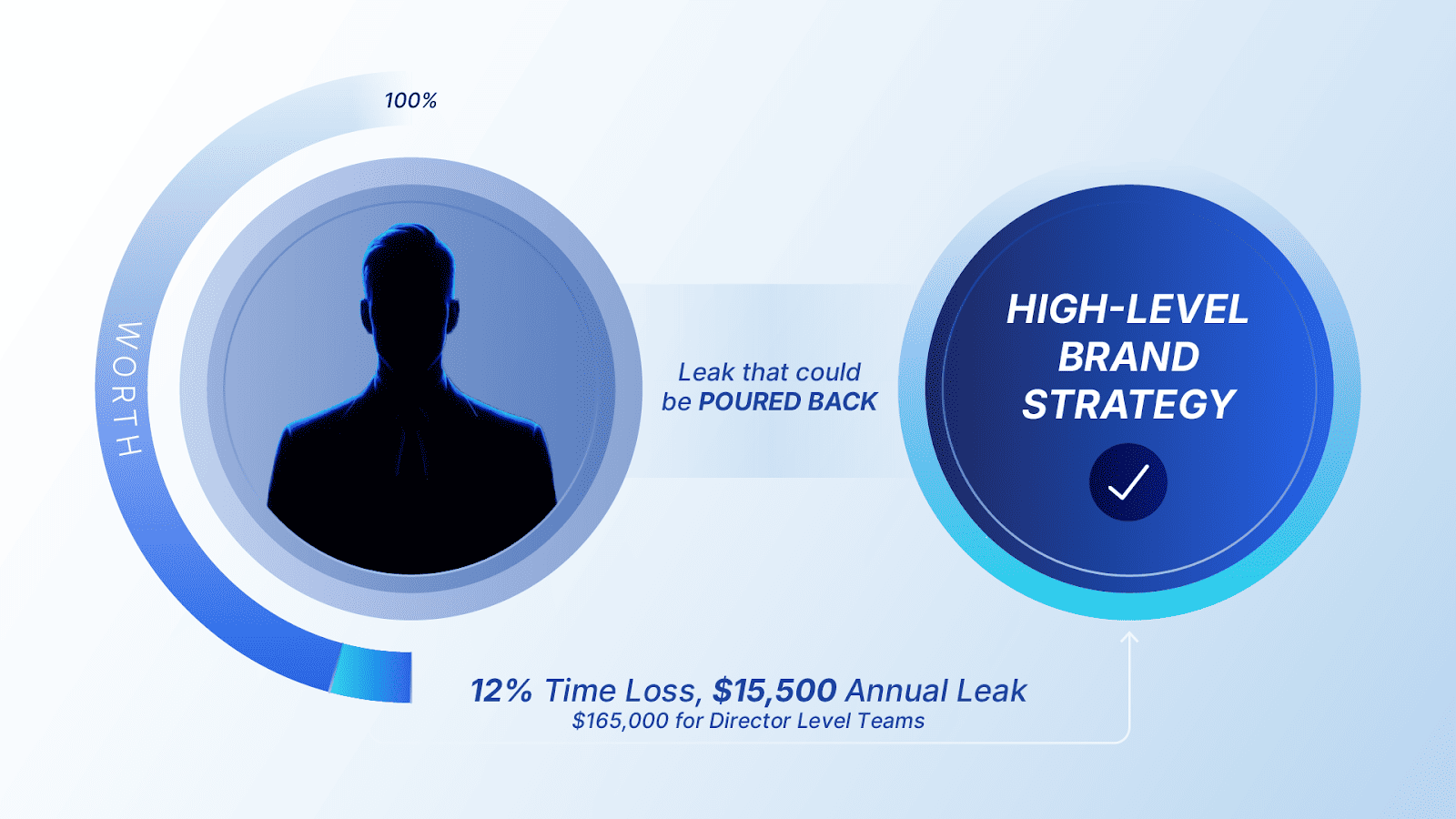
The Final ROI: Redirecting Hours into High-Level Strategy
The final shift for the forward-thinking Brand Director is moving from a "Data Dump" mindset toward true Clinical Authority.
By eliminating executive drag, organizations can transform their pharmaceutical presentation templates from simple assets into high-velocity strategic tools.
- Individual Financial Recovery: By unbundling design production from high-overhead agencies, organizations redirect approximately $15,500 per year, per manager, back into the science that drives market share.
- The Director Level Impact: For a team of senior leaders, this represents a reclaimable Strategic Bandwidth valued at over $165,000 in executive time annually.
- Maximizing Operational Surplus: When accounting for the hidden costs of manual labor, pharma brand management efficiency becomes the most significant driver of a lean launch budget.
- Audit-Ready Scale: Utilizing specialized PowerPoint themes for scientific presentations ensures that every pharma product slide remains compliant as you scale for global congresses.





